
Attempting a demonstration with hydrogen peroxide, The Professor stumbles over an alternative!
- Subject:
- Chemistry
- Physical Science
- Material Type:
- Lesson
- Date Added:
- 10/31/2013

Attempting a demonstration with hydrogen peroxide, The Professor stumbles over an alternative!
Paul Andersen explains how acid-base chemistry can be understood in terms of equilibrium. Water is present in all acid-base chemistry and is amphoteric in nature. The Ka and Kb values can be used to determine the strength of an acid or a base. Titrations can be used to student neutralization reactions between strong and weak acids and bases.

The video resource "Acid-Base Reactions in Solution: Crash Course Chemistry #8" is included in the "Chemistry" course from the resources series of "Crash Course". Crash Course is a educational video series from John and Hank Green.
Using acid-base titration to find mass of oxalic acid
Acetic Acid to Acetyl Chloride mechanism. Can be generalized to forming any acid halide from a carboxylic acid
Paul Andersen explains pH as the power of hydrogen. He explains how increases in the hydronium ion (or hydrogen ion) concentration can lower the pH and create acids. He also explains how the reverse is true. An analysis of a strong acid and strong base is also included.

Actinium is element number 89, and first of the actinide series. This video is one of the 118 clips included in the periodic table of elements themed collection created by Brady Haran and the University of Nottingham in the UK.
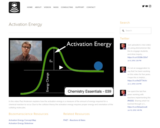
In this video Paul Andersen explains how the activation energy is a measure of the amount of energy required for a chemical reaction to occur. Due to the collision theory the activation energy requires proper energy and orientation of the colliding molecules.
Alcohol Properties
Naming alcohols
Aldehyde introduction
Aldol Reaction
This video is from the Khan Academy subject of Science on the topic of Organic chemistry and it covers Alkene intro and stability.
This video is from the Khan Academy subject of Science on the topic of Organic chemistry and it covers Alkene nomenclature.

The video resource "Alkenes & Alkynes - Crash Course Chemistry #41" is included in the "Chemistry" course from the resources series of "Crash Course". Crash Course is a educational video series from John and Hank Green.
Is climate change real? Yes, it is! And technologies to reduce Greenhouse Gas (GHG) emissions are being developed. One type of technology that is imperative in the short run is biofuels; however, biofuels must meet specifications for gasoline, diesel, and jet fuel, or catastrophic damage could occur. This course will examine the chemistry of technologies of bio-based sources for power generation and transportation fuels. We'll consider various biomasses that can be utilized for fuel generation, understand the processes necessary for biomass processing, explore biorefining, and analyze how biofuels can be used in current fuel infrastructure.

Aluminium is element 13 on the periodic table. This video is one of the 118 clips included in the periodic table of elements themed collection created by Brady Haran and the University of Nottingham in the UK.

Students learn how nanoparticles can be creatively used for medical diagnostic purposes. They learn about buckminsterfullerenes, more commonly known as buckyballs, and about the potential for these complex carbon molecules to deliver drugs and other treatments into the human body. They brainstorm methods to track buckyballs in the body, then build a buckyball from pipe cleaners with a fluorescent tag to model how nanoparticles might be labeled and detected for use in a living organism. As an extension, students research and select appropriate radioisotopes for different medical applications.

Americium is element number 95. This video is one of the 118 clips included in the periodic table of elements themed collection created by Brady Haran and the University of Nottingham in the UK.
Amide Formation from Acyl Chloride